THERAPEUTICS PIPELINE
Plantar Fasciitis
Plantar fasciitis is a painful and sometimes debilitating condition caused by the inflammation of the tissue that runs across the bottom of the foot and connects a heel bone to the toes. The pain from plantar fasciitis can become excruciating and chronic that makes work, exercise and ordinary walking challenging and difficult.1
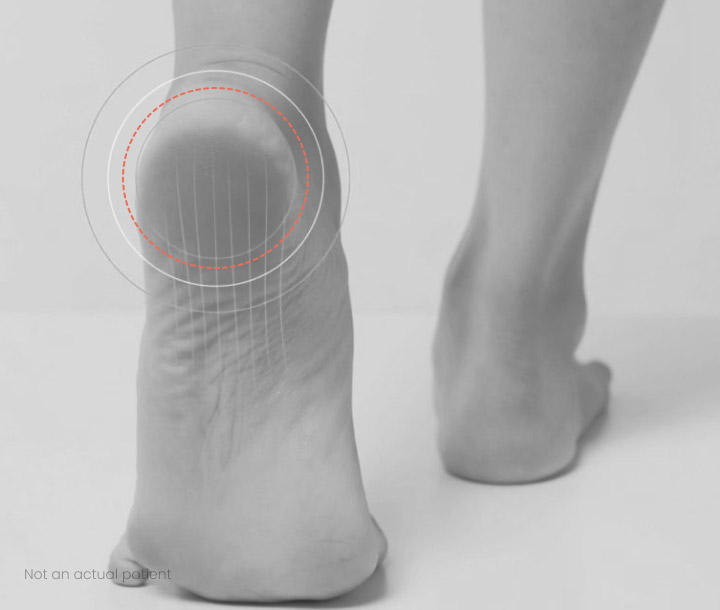
In November 2020, Revance announced results from a Phase 2 clinical trial evaluating DAXXIFY® for Injection for the management of plantar fasciitis. Revance plans to analyze the data further for additional insights, however our primary focus will be on neurological indications, where the market and path to approval are well established.
revance
Plantar Fasciitis in Numbers
Will suffer in U.S.
Experience Symptoms
Symptoms Last
What is Plantar Fasciitis? With Dr. Walter Strash

Unmet Need

Beyond plantar fasciitis, Revance is continuing to study DaxibotulinumtoxinA for Injection in other important therapeutic indications with a current focus in muscle movement disorders.
DaxibotulinumtoxinA for Injection has delivered highly positive Phase 3 results, including strong duration of effect, demonstrating a median duration of 24 weeks in the treatment of glabellar lines and cervical dystonia.
References
1. Mayo Clinic Web Site. https://www.mayoclinic.org/diseases-conditions/plantar-fasciitis/symptoms-causes/syc-20354846. Accessed 8/11/20
2. Schneider H. et al. ACFS Clinical Consenus Statement. Journal of Foot and Ankle Surgery 2017
3. Tehririan MA et al. Plantar Fasciitis. J Res Med Sci. 2012 Aug; 17(8): 799–804
IMPORTANT SAFETY INFORMATION for DAXXIFY® (daxibotulinumtoxinA-Ianm) injection
DAXXIFY® may cause serious side effects that can be life threatening.
DAXXIFY® (daxibotulinumtoxinA-lanm) injection IMPORTANT SAFETY INFORMATION
Indications
(daxibotulinumtoxinA-lanm) injection is an acetylcholine release inhibitor and neuromuscular blocking agent indicated for the temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity in adult patients and for the treatment of cervical dystonia in adults.
WARNING: DISTANT SPREAD OF TOXIN EFFECT The effects of DAXXIFY® and all botulinum toxin products may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life threatening and there have been reports of death. DAXXIFY® is not approved for the treatment of spasticity or any conditions other than cervical dystonia and glabellar lines.
IMPORTANT SAFETY INFORMATION
Contraindications
DAXXIFY® contraindications include hypersensitivity to any botulinum toxin preparation or any of the components in the formulation and infection at the injection site(s).
Warnings and Precautions
Please refer to Boxed Warning for Distant Spread of Toxin Effect.
The potency Units of DAXXIFY® are not interchangeable with other preparations of other botulinum toxin products. Recommended dose and frequency of administration should not be exceeded. Patients should seek immediate medical attention if respiratory, speech or swallowing difficulties occur. Use caution when administering to patients with pre-existing cardiovascular disease. Concomitant neuromuscular disorders may exacerbate clinical effects of treatment.
Adverse Reactions
The most commonly observed adverse reactions are:
Glabeller lines (≥1%) were headache (6%), eyelid ptosis (2%) and facial paresis (1%).
Cervical Dystonia (≥5%): headache (9%), injection site pain (8%), injection site erythema (5%), muscular weakness (5%), and upper respiratory tract infection (5%).
Drug Interactions
Co-administration of DAXXIFY® and aminoglycoside antibiotics, anticholinergic agents or any other agents interfering with neuromuscular transmission or muscle relaxants should only be performed with caution as the effect of DAXXIFY® may be potentiated. The effect of administering different botulinum neurotoxins during course of treatment with DAXXIFY® is unknown.
Use in Specific Populations
DAXXIFY® is not recommended for use in children or pregnant women.
Please see DAXXIFY® full Prescribing Information, including Boxed Warning and Medication Guide.
To report side effects associated with DAXXIFY®, please visit safety.revance.com, or call 1-877-373-8669. You may also report side effects to the FDA at 1-800-FDA-1088 or visit www.fda.gov/medwatch
DAXI-004726